
Wernig Laboratory
October 9, 2024: Welcome new postdoc Kirill to the lab!
Sep 17, 2024: Jinzhao's paper accepted in principle!
Jul 11, 2024: Gernot's paper published in Nature Communications!
March 21, 2024: Marius Mader's paper comes out in Nature Neuroscience!
Oct 10, 2023: Yongjin receives the Sammy Kuo Award from the Neuroscience Institute - CONGRATULATIONS!
Yongjin's paper on cell therapy in a mouse AD model is published in Cell Stem Cells
Our lab is generally interested in the molecular mechanisms that determine cell fates
Recently, we have identified a pool of transcription factors that are sufficient to convert skin fibroblasts directly into functional neuronal cells that we termed induced neuronal (iN) cells. This was a surprising finding and indicated that direct lineage reprogramming may be applicable to many somatic cell types and many different directions. Indeed, following our work others have identified transcription factors that could induce cardiomyocytes, blood progenitors, and hepatocytes from fibroblasts.
We are now focussing on two major aspects of iN and iPS cell reprogramming:
(i) we are fascinated by the puzzle how a hand full of transcription factors can so efficiently reprogram the entire epigenome of a cell so that it changes identity. To that end we are applying genome-wide expression analysis, chromatin immunoprecipitation, protein biochemistry, proteomics and functional screens.
(ii) it is equally exciting to now use reprogramming methods as tools to study or treat certain diseases. iPS cells have the great advantage that they can easily be genetically manipulated rendering them ideal for treating monogenetic disorders when combined with cell transplantation-based therapies. In particular we are working on Dystrophic Epidermolysis Bullosa in collaboration with Stanford's Dermatology Department. An exciting application of iN cell technology will be to try modeling neurological diseases in vitro. We perform both mouse and human experiments hoping to identify quantifiable phenotypes correlated with genotype and in a second step evaluate whether this assay could be used to discover novel drugs improve the disease progression.
Wernig Lab Research
Overview
Our lab is interested in the molecular mechanisms that define neural lineage identity focusing on transcription factors and chromatin biology. We use cellular reprogramming to understand how neurons are induced, how they mature and maintain their identity. Reprogramming also allows us to generate a novel tool box to study human neuronal and glial cell biology which become powerful human disease models in combination with genetic engineering. We further seek to develop reprogramming & genetic engineering approaches towards stem cell-based therapies. Finally, we study microglia-neuron interactions with the ultimate goal to understand the brain's immune system in health and disease and to exploit microglia for therapeutic and regenerative purposes.
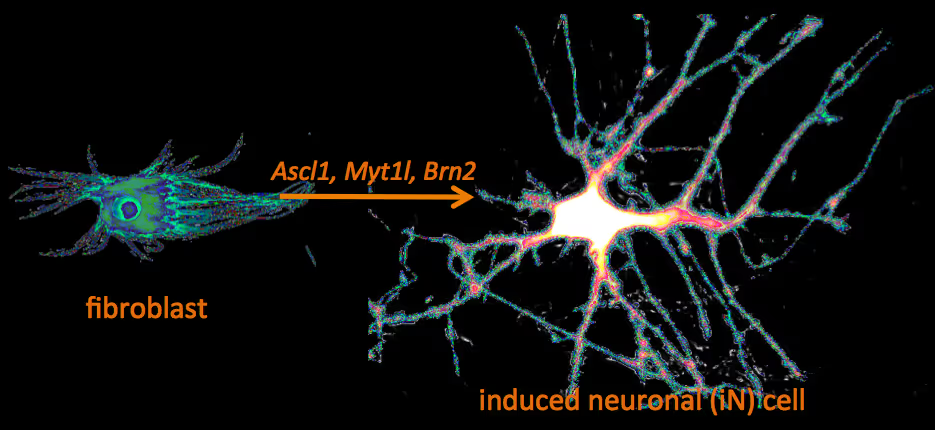
Human neuronal cell disease modeling
Neurosychiatric diseases like autism and schizophrenia are highly complex brain disorders difficult to model in mice in part due to complex genetic etiology and sometimes affecting human-specific genes. We develop novel human cell models to investigate disease-relevant cell biological phenomena.
Generation of defined human neuronal cell types to study neuronal cell biology
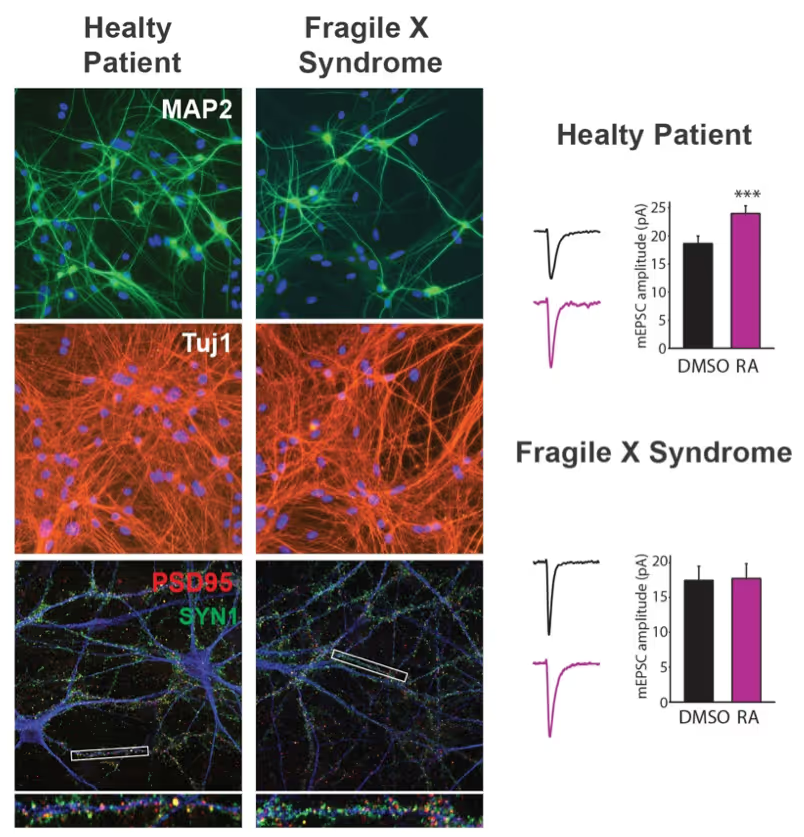
We have and continue to develop protocols to generate specific types of neurons such as pure glutamatergic and pure GABAergic neurons from human pluripotent stem cells using transcription factors. In combination with genetic engineering or deriving iPS cells from patients, we then interrogate the cell biology of human neurons that carry disease-causing mutations. A particular focus is on synaptic function as shown in the figure on the right on Fragile X Syndrome neurons in collaboration with Lu Chen and Tom Südhof's laboratories.
Making neurons from blood
The ability to generate functional induced neuronal cells from distantly related somatic cell types is fascinating but also offers the opportunity to obtain neurons from a larger cohort of human subjects. In particular blood is readily available and we showed can be efficiently converted into functional neurons from young and aged donors.
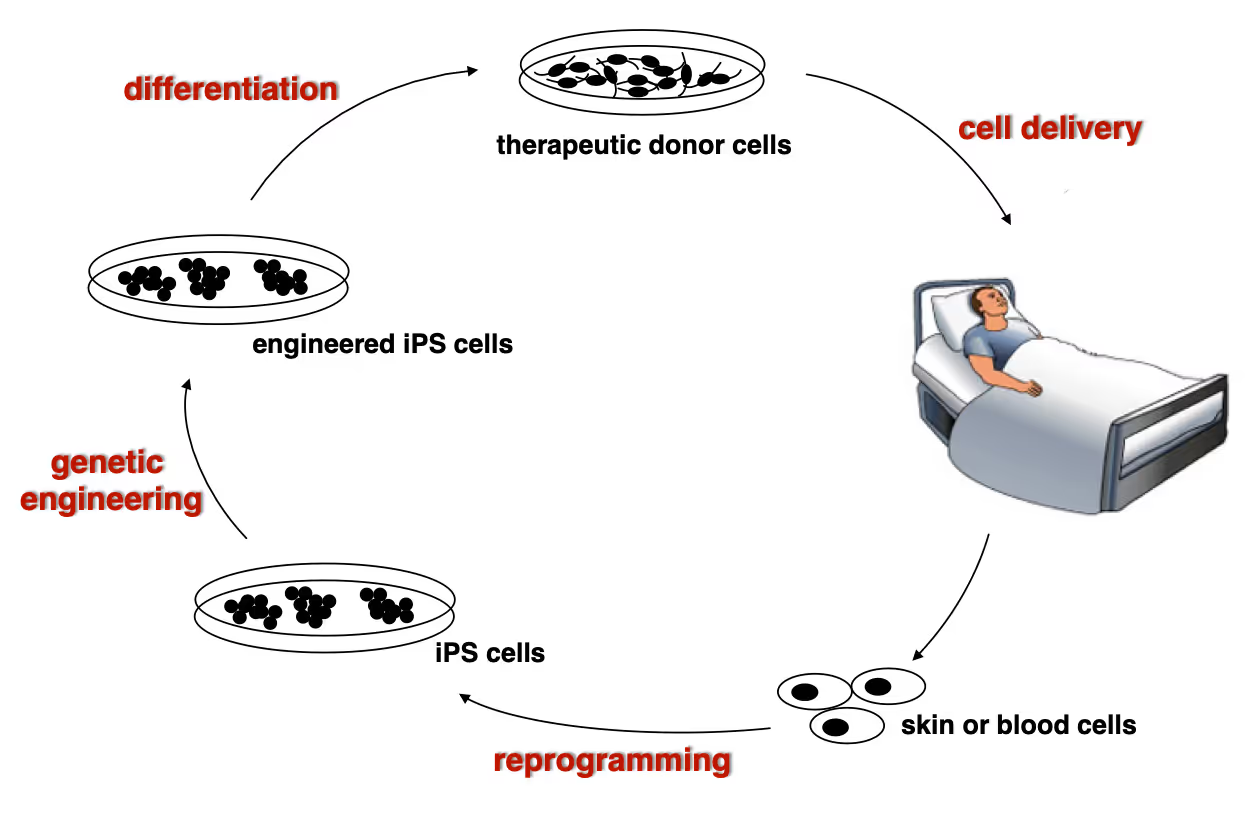
Developing next generation cell therapies
The combination of reprogramming and gene editing is truly powerful as it provides exciting new possibilities to generate cells that can be transplanted and have disease modifying activity. We currently apply this approach to restore mono-genetic diseases, but our vision goes beyond simple regenerative medicine. We will be able to genetically engineer designer cells that functionally integrate into diseased tissue equipped with sensing and intelligent disease-response mechanisms.
Towards a Phase 1 clinical trial for the fatal skin disease Epidermolysis Bullosa
Dystrophic Epidermolysis Bullosa is a severe, blistering monogenetic skin disease caused by mutations in the gene coding for type VII collagen. We have developed a 1-step gene editing/iPS cell reprogramming method to rapidly generate patient iPS cells corrected for their disease-causing mutations in the Collagen7a1 gene. In collaboration with dermatologist Tony Oro we are developing a cell manufacturing process compatible with Good Manufacturing Procedures (GMP) to obtain FDA-approval for a first in man Phase I clinical trial with with a genetically engineered iPS cell product.
Exploiting glia cell transplantation to treat neurodegenerative disease
Both oligodendrocyte precursor cells as well as microglia can efficiently repopulate the brain. We are interested in exploiting the properties of these cells to develop novel cell therapies for the brain either to use the transplanted cells to restore function such as myelination, to alter the function of transplanted cells for therepeutic benefit, to use the cells as vehicles for therapeutic molecules, or ultimately to develop designer cells that are engineered with genetic synthetic biology circuits to sense and interfere with disease processes of the brain.
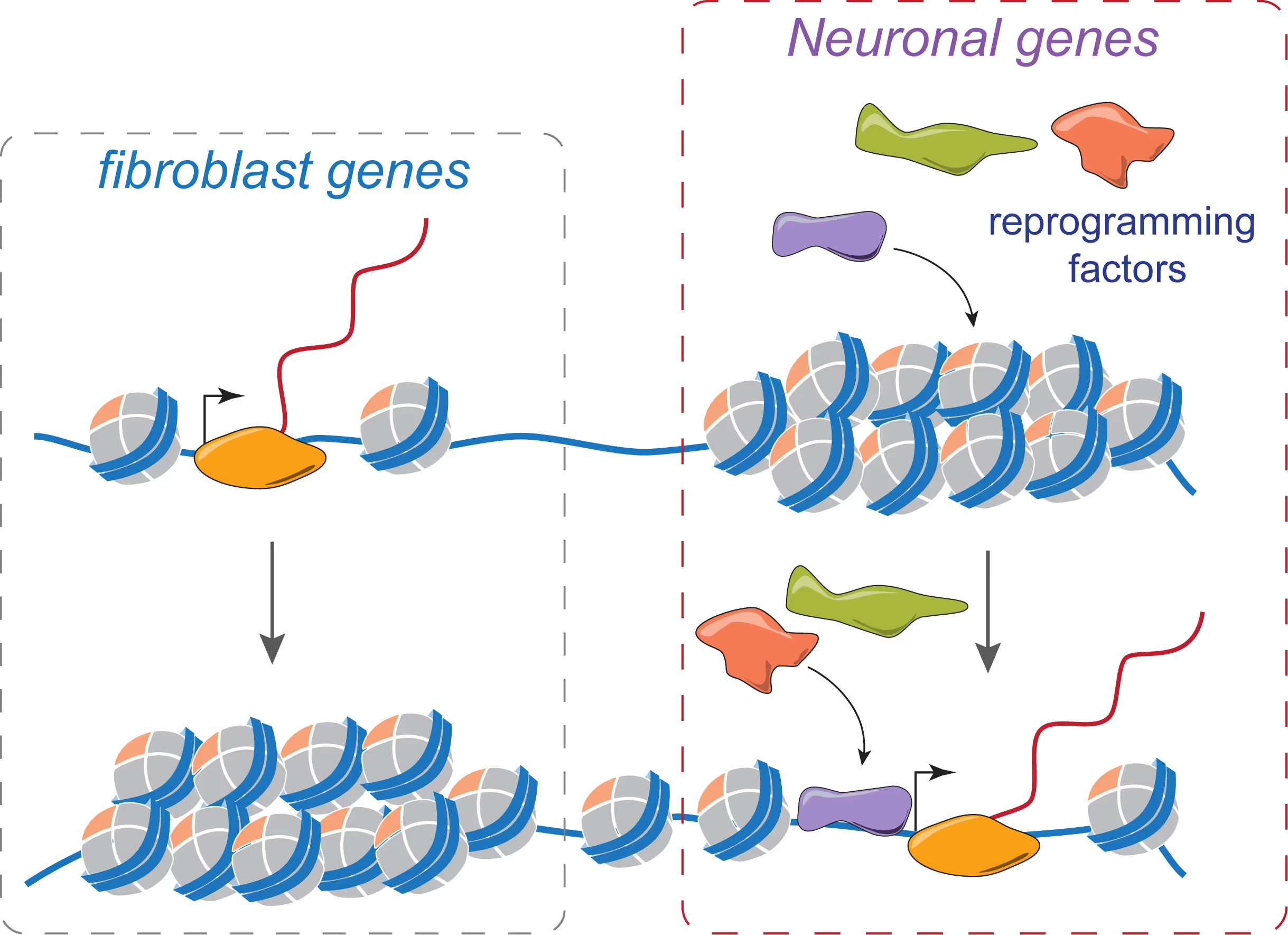
Mechanisms of neural cell lineage identity
We are interested in the molecular mechanisms that define neuronal and glial cell identity. We found sets of transcription factors that can convert fibroblasts or lymphocytes into neurons and oligodendrocytes. These factors are also operational during normal development and are largely responsible to induce terminal lineages from progenitor cells.
"On target" pioneer factors and chromatin remodeling during neuronal induction
We found that Ascl1, one of our reprogramming factors, has a unique ability to access its physiological targets even in fibroblasts where these sites are in a closed chromatin configuration. We are fascinated by this "on target" pioneering property and are investigating how Ascl1 can access its target sites in an unfavorable chromatin environment and how it then remodels the chromatin at these sites to activate the neuronal transcriptional program.
Maintenance of neuronal identity
Once neurons are made, there ought to be also mechanisms that maintain neuronal identity. We stumbled upon a novel repressive mechanism: The neuronal-specific transcription factor Myt1l continuosly represses many non-neuronal programs in neurons leaving the neuronal program open to activate by other factors and thereby ensuring stable neuronal gene expression. Myt1l was also recently found to be mutated in autism and schizophrenia.
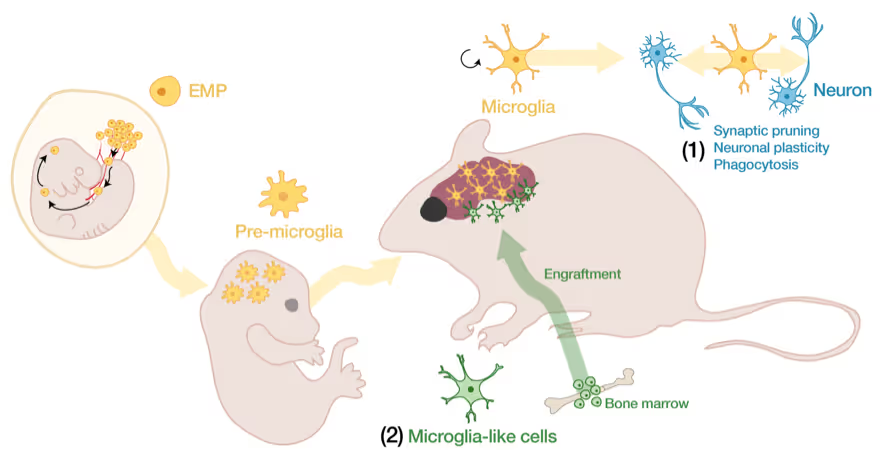
Microglia-neuron interactions in the healthy and diseased brain
Microglia, the brain's resident immune cells, are fascinating cells. They are derived from yolk sac progenitor cells early during development, are long-lived, and are not exchanged from bone marrow progenitor cells under physiological conditions. Microglia have been implicated in synaptic pruning, adult neurogenesis, and various brain diseases including Alzheimer's disease and Schizophrenia.
Developing an efficient microglia replacement system
We have developed a method to efficiently replace endogenous microglia from circulating cells without genetic manipulation. This does not happen physiologically but under certain conditions peripheral blood cells cross the blood-brain-barrier, migrate into the brain parenchyma and replace endogenous cells. We are investigating the cellular and molecular signals that enable circulating cells to invade the brain in order to further improve microglia replacement strategies.
The role of microglia in the normal and the diseased brain
Our ability to replace microglia provides us with a powerful tool to functionally perturb microglia function in normal and disease states. E.g. the microglial gene TREM2 is a strong Alzheimer's disease risk gene, but major questions about the neuro-immune interplay in the context of neurodegeneration and aging remain unsolved. Microglia replacement also provides an exciting prospect to develop novel cell therapies for a variety of brain diseases including enzyme deficiency syndromes, neurodegeneration, and brain tumors.
Lab Gene Expression Data
Publications
De Los Angeles A, Ferrari F, Fujiwara Y, Mathieu R, Lee S, Lee S, Tu HC, Ross S, Chou S, Nguyen M, Wu Z, Theunissen TW, Powell BE, Imsoonthornruksa S, Chen J, Borkent M, Krupalnik V, Lujan E, Wernig M, Hanna JH, Hochedlinger K, Pei D, Jaenisch R, Deng H, Orkin SH, Park PJ, Daley GQ
Tanabe K, Haag D, Wernig M
The predominant view of embryonic development and cell differentiation has been that rigid and even irreversible epigenetic marks are laid down along the path of cell specialization ensuring the proper silencing of unrelated lineage programmes. This model made the prediction that specialized cell types are stable and cannot be redirected into other lineages. Accordingly, early attempts to change the identity of somatic cells had little success and was limited to conversions between closely related cell types. Nuclear transplantation experiments demonstrated, however, that specialized cells even from adult mammals can be reprogrammed into a totipotent state. The discovery that a small combination of transcription factors can reprogramme cells to pluripotency without the need of oocytes further supported the view that these epigenetic barriers can be overcome much easier than assumed, but the extent of this flexibility was still unclear. When we showed that a differentiated mesodermal cell can be directly converted to a differentiated ectodermal cell without a pluripotent intermediate, it was suggested that in principle any cell type could be converted into any other cell type. Indeed, the work of several groups in recent years has provided many more examples of direct somatic lineage conversions. Today, the question is not anymore whether a specific cell type can be generated by direct reprogramming but how it can be induced.
The predominant view of embryonic development and cell differentiation has been that rigid and even irreversible epigenetic marks are laid down along the path of cell specialization ensuring the proper silencing of unrelated lineage programmes. This model made the prediction that specialized cell types are stable and cannot be redirected into other lineages. Accordingly, early attempts to change the identity of somatic cells had little success and was limited to conversions between closely related cell types. Nuclear transplantation experiments demonstrated, however, that specialized cells even from adult mammals can be reprogrammed into a totipotent state. The discovery that a small combination of transcription factors can reprogramme cells to pluripotency without the need of oocytes further supported the view that these epigenetic barriers can be overcome much easier than assumed, but the extent of this flexibility was still unclear. When we showed that a differentiated mesodermal cell can be directly converted to a differentiated ectodermal cell without a pluripotent intermediate, it was suggested that in principle any cell type could be converted into any other cell type. Indeed, the work of several groups in recent years has provided many more examples of direct somatic lineage conversions. Today, the question is not anymore whether a specific cell type can be generated by direct reprogramming but how it can be induced.
Kareta MS, Sage J, Wernig M
Pluripotent stem cells, defined by an unlimited self-renewal capacity and an undifferentiated state, are best typified by embryonic stem cells. These cells have a unique cell cycle compared to somatic cells as defined by a rapid progression through the cell cycle and a minimal time spent in G1. Recent reports indicate that pluripotency and cell cycle regulation are mechanistically linked. In this review, we discuss the reciprocal co-regulation of these processes, how this co-regulation may prevent differentiation, and how cellular reprogramming can re-establish the unique cell cycle regulation in induced pluripotent stem cells.
Pluripotent stem cells, defined by an unlimited self-renewal capacity and an undifferentiated state, are best typified by embryonic stem cells. These cells have a unique cell cycle compared to somatic cells as defined by a rapid progression through the cell cycle and a minimal time spent in G1. Recent reports indicate that pluripotency and cell cycle regulation are mechanistically linked. In this review, we discuss the reciprocal co-regulation of these processes, how this co-regulation may prevent differentiation, and how cellular reprogramming can re-establish the unique cell cycle regulation in induced pluripotent stem cells.
Patzke C, Han Y, Covy J, Yi F, Maxeiner S, Wernig M, Südhof TC
Heterozygous mutations in the syntaxin-binding protein 1 (STXBP1) gene, which encodes Munc18-1, a core component of the presynaptic membrane-fusion machinery, cause infantile early epileptic encephalopathy (Ohtahara syndrome), but it is unclear how a partial loss of Munc18-1 produces this severe clinical presentation. Here, we generated human ES cells designed to conditionally express heterozygous and homozygous STXBP1 loss-of-function mutations and studied isogenic WT and STXBP1-mutant human neurons derived from these conditionally mutant ES cells. We demonstrated that heterozygous STXBP1 mutations lower the levels of Munc18-1 protein and its binding partner, the t-SNARE-protein Syntaxin-1, by approximately 30% and decrease spontaneous and evoked neurotransmitter release by nearly 50%. Thus, our results confirm that using engineered human embryonic stem (ES) cells is a viable approach to studying disease-associated mutations in human neurons on a controlled genetic background, demonstrate that partial STXBP1 loss of function robustly impairs neurotransmitter release in human neurons, and suggest that heterozygous STXBP1 mutations cause early epileptic encephalopathy specifically through a presynaptic impairment.
Heterozygous mutations in the syntaxin-binding protein 1 (STXBP1) gene, which encodes Munc18-1, a core component of the presynaptic membrane-fusion machinery, cause infantile early epileptic encephalopathy (Ohtahara syndrome), but it is unclear how a partial loss of Munc18-1 produces this severe clinical presentation. Here, we generated human ES cells designed to conditionally express heterozygous and homozygous STXBP1 loss-of-function mutations and studied isogenic WT and STXBP1-mutant human neurons derived from these conditionally mutant ES cells. We demonstrated that heterozygous STXBP1 mutations lower the levels of Munc18-1 protein and its binding partner, the t-SNARE-protein Syntaxin-1, by approximately 30% and decrease spontaneous and evoked neurotransmitter release by nearly 50%. Thus, our results confirm that using engineered human embryonic stem (ES) cells is a viable approach to studying disease-associated mutations in human neurons on a controlled genetic background, demonstrate that partial STXBP1 loss of function robustly impairs neurotransmitter release in human neurons, and suggest that heterozygous STXBP1 mutations cause early epileptic encephalopathy specifically through a presynaptic impairment.
Zunder ER, Lujan E, Goltsev Y, Wernig M, Nolan GP
To analyze cellular reprogramming at the single-cell level, mass cytometry was used to simultaneously measure markers of pluripotency, differentiation, cell-cycle status, and cellular signaling throughout the reprogramming process. Time-resolved progression analysis of the resulting data sets was used to construct a continuous molecular roadmap for three independent reprogramming systems. Although these systems varied substantially in Oct4, Sox2, Klf4, and c-Myc stoichiometry, they presented a common set of reprogramming landmarks. Early in the reprogramming process, Oct4(high)Klf4(high) cells transitioned to a CD73(high)CD104(high)CD54(low) partially reprogrammed state. Ki67(low) cells from this intermediate population reverted to a MEF-like phenotype, but Ki67(high) cells advanced through the M-E-T and then bifurcated into two distinct populations: an ESC-like Nanog(high)Sox2(high)CD54(high) population and a mesendoderm-like Nanog(low)Sox2(low)Lin28(high)CD24(high)PDGFR-α(high) population. The methods developed here for time-resolved, single-cell progression analysis may be used for the study of additional complex and dynamic systems, such as cancer progression and embryonic development.
To analyze cellular reprogramming at the single-cell level, mass cytometry was used to simultaneously measure markers of pluripotency, differentiation, cell-cycle status, and cellular signaling throughout the reprogramming process. Time-resolved progression analysis of the resulting data sets was used to construct a continuous molecular roadmap for three independent reprogramming systems. Although these systems varied substantially in Oct4, Sox2, Klf4, and c-Myc stoichiometry, they presented a common set of reprogramming landmarks. Early in the reprogramming process, Oct4(high)Klf4(high) cells transitioned to a CD73(high)CD104(high)CD54(low) partially reprogrammed state. Ki67(low) cells from this intermediate population reverted to a MEF-like phenotype, but Ki67(high) cells advanced through the M-E-T and then bifurcated into two distinct populations: an ESC-like Nanog(high)Sox2(high)CD54(high) population and a mesendoderm-like Nanog(low)Sox2(low)Lin28(high)CD24(high)PDGFR-α(high) population. The methods developed here for time-resolved, single-cell progression analysis may be used for the study of additional complex and dynamic systems, such as cancer progression and embryonic development.
Batista PJ, Molinie B, Wang J, Qu K, Zhang J, Li L, Bouley DM, Lujan E, Haddad B, Daneshvar K, Carter AC, Flynn RA, Zhou C, Lim KS, Dedon P, Wernig M, Mullen AC, Xing Y, Giallourakis CC, Chang HY
N6-methyl-adenosine (m(6)A) is the most abundant modification on messenger RNAs and is linked to human diseases, but its functions in mammalian development are poorly understood. Here we reveal the evolutionary conservation and function of m(6)A by mapping the m(6)A methylome in mouse and human embryonic stem cells. Thousands of messenger and long noncoding RNAs show conserved m(6)A modification, including transcripts encoding core pluripotency transcription factors. m(6)A is enriched over 3' untranslated regions at defined sequence motifs and marks unstable transcripts, including transcripts turned over upon differentiation. Genetic inactivation or depletion of mouse and human Mettl3, one of the m(6)A methylases, led to m(6)A erasure on select target genes, prolonged Nanog expression upon differentiation, and impaired ESC exit from self-renewal toward differentiation into several lineages in vitro and in vivo. Thus, m(6)A is a mark of transcriptome flexibility required for stem cells to differentiate to specific lineages.
N6-methyl-adenosine (m(6)A) is the most abundant modification on messenger RNAs and is linked to human diseases, but its functions in mammalian development are poorly understood. Here we reveal the evolutionary conservation and function of m(6)A by mapping the m(6)A methylome in mouse and human embryonic stem cells. Thousands of messenger and long noncoding RNAs show conserved m(6)A modification, including transcripts encoding core pluripotency transcription factors. m(6)A is enriched over 3' untranslated regions at defined sequence motifs and marks unstable transcripts, including transcripts turned over upon differentiation. Genetic inactivation or depletion of mouse and human Mettl3, one of the m(6)A methylases, led to m(6)A erasure on select target genes, prolonged Nanog expression upon differentiation, and impaired ESC exit from self-renewal toward differentiation into several lineages in vitro and in vivo. Thus, m(6)A is a mark of transcriptome flexibility required for stem cells to differentiate to specific lineages.
Sebastiano V, Zhen HH, Haddad B, Derafshi BH, Bashkirova E, Melo SP, Wang P, Leung TL, Siprashvili Z, Tichy A, Li J, Ameen M, Hawkins J, Lee S, Li L, Schwertschkow A, Bauer G, Lisowski L, Kay MA, Kim SK, Lane AT, Wernig M, Oro AE
Patients with recessive dystrophic epidermolysis bullosa (RDEB) lack functional type VII collagen owing to mutations in the gene COL7A1 and suffer severe blistering and chronic wounds that ultimately lead to infection and development of lethal squamous cell carcinoma. The discovery of induced pluripotent stem cells (iPSCs) and the ability to edit the genome bring the possibility to provide definitive genetic therapy through corrected autologous tissues. We generated patient-derived COL7A1-corrected epithelial keratinocyte sheets for autologous grafting. We demonstrate the utility of sequential reprogramming and adenovirus-associated viral genome editing to generate corrected iPSC banks. iPSC-derived keratinocytes were produced with minimal heterogeneity, and these cells secreted wild-type type VII collagen, resulting in stratified epidermis in vitro in organotypic cultures and in vivo in mice. Sequencing of corrected cell lines before tissue formation revealed heterogeneity of cancer-predisposing mutations, allowing us to select COL7A1-corrected banks with minimal mutational burden for downstream epidermis production. Our results provide a clinical platform to use iPSCs in the treatment of debilitating genodermatoses, such as RDEB.
Patients with recessive dystrophic epidermolysis bullosa (RDEB) lack functional type VII collagen owing to mutations in the gene COL7A1 and suffer severe blistering and chronic wounds that ultimately lead to infection and development of lethal squamous cell carcinoma. The discovery of induced pluripotent stem cells (iPSCs) and the ability to edit the genome bring the possibility to provide definitive genetic therapy through corrected autologous tissues. We generated patient-derived COL7A1-corrected epithelial keratinocyte sheets for autologous grafting. We demonstrate the utility of sequential reprogramming and adenovirus-associated viral genome editing to generate corrected iPSC banks. iPSC-derived keratinocytes were produced with minimal heterogeneity, and these cells secreted wild-type type VII collagen, resulting in stratified epidermis in vitro in organotypic cultures and in vivo in mice. Sequencing of corrected cell lines before tissue formation revealed heterogeneity of cancer-predisposing mutations, allowing us to select COL7A1-corrected banks with minimal mutational burden for downstream epidermis production. Our results provide a clinical platform to use iPSCs in the treatment of debilitating genodermatoses, such as RDEB.
Ang CE, Wernig M
Cellular differentiation processes during normal embryonic development are guided by extracellular soluble factors such as morphogen gradients and cell contact signals, eventually resulting in induction of specific combinations of lineage-determining transcription factors. The young field of epigenetic reprogramming takes advantage of this knowledge and uses cell fate determination factors to convert one lineage into another such as the conversion of fibroblasts into pluripotent stem cells or neurons. These induced cell fate conversions open up new avenues for studying disease processes, generating cell material for therapeutic intervention such as drug screening and potentially also for cell-based therapies. However, there are still limitations that have to be overcome to fulfill these promises, centering on reprogramming efficiencies, cell identity, and maturation. In this review, we discuss the discovery of induced neuronal reprogramming, ways to improve the conversion process, and finally how to define properly the identity of those converted neuronal cells.
Cellular differentiation processes during normal embryonic development are guided by extracellular soluble factors such as morphogen gradients and cell contact signals, eventually resulting in induction of specific combinations of lineage-determining transcription factors. The young field of epigenetic reprogramming takes advantage of this knowledge and uses cell fate determination factors to convert one lineage into another such as the conversion of fibroblasts into pluripotent stem cells or neurons. These induced cell fate conversions open up new avenues for studying disease processes, generating cell material for therapeutic intervention such as drug screening and potentially also for cell-based therapies. However, there are still limitations that have to be overcome to fulfill these promises, centering on reprogramming efficiencies, cell identity, and maturation. In this review, we discuss the discovery of induced neuronal reprogramming, ways to improve the conversion process, and finally how to define properly the identity of those converted neuronal cells.
Chanda S, Ang CE, Davila J, Pak C, Mall M, Lee QY, Ahlenius H, Jung SW, Südhof TC, Wernig M
Direct conversion of nonneural cells to functional neurons holds great promise for neurological disease modeling and regenerative medicine. We previously reported rapid reprogramming of mouse embryonic fibroblasts (MEFs) into mature induced neuronal (iN) cells by forced expression of three transcription factors: ASCL1, MYT1L, and BRN2. Here, we show that ASCL1 alone is sufficient to generate functional iN cells from mouse and human fibroblasts and embryonic stem cells, indicating that ASCL1 is the key driver of iN cell reprogramming in different cell contexts and that the role of MYT1L and BRN2 is primarily to enhance the neuronal maturation process. ASCL1-induced single-factor neurons (1F-iN) expressed mature neuronal markers, exhibited typical passive and active intrinsic membrane properties, and formed functional pre- and postsynaptic structures. Surprisingly, ASCL1-induced iN cells were predominantly excitatory, demonstrating that ASCL1 is permissive but alone not deterministic for the inhibitory neuronal lineage.
Direct conversion of nonneural cells to functional neurons holds great promise for neurological disease modeling and regenerative medicine. We previously reported rapid reprogramming of mouse embryonic fibroblasts (MEFs) into mature induced neuronal (iN) cells by forced expression of three transcription factors: ASCL1, MYT1L, and BRN2. Here, we show that ASCL1 alone is sufficient to generate functional iN cells from mouse and human fibroblasts and embryonic stem cells, indicating that ASCL1 is the key driver of iN cell reprogramming in different cell contexts and that the role of MYT1L and BRN2 is primarily to enhance the neuronal maturation process. ASCL1-induced single-factor neurons (1F-iN) expressed mature neuronal markers, exhibited typical passive and active intrinsic membrane properties, and formed functional pre- and postsynaptic structures. Surprisingly, ASCL1-induced iN cells were predominantly excitatory, demonstrating that ASCL1 is permissive but alone not deterministic for the inhibitory neuronal lineage.
Cho A, Tang Y, Davila J, Deng S, Chen L, Miller E, Wernig M, Graef IA
Development of the nervous system begins with neural induction, which is controlled by complex signaling networks functioning in concert with one another. Fine-tuning of the bone morphogenetic protein (BMP) pathway is essential for neural induction in the developing embryo. However, the molecular mechanisms by which cells integrate the signaling pathways that contribute to neural induction have remained unclear. We find that neural induction is dependent on the Ca(2+)-activated phosphatase calcineurin (CaN). Fibroblast growth factor (FGF)-regulated Ca(2+) entry activates CaN, which directly and specifically dephosphorylates BMP-regulated Smad1/5 proteins. Genetic and biochemical analyses revealed that CaN adjusts the strength and transcriptional output of BMP signaling and that a reduction of CaN activity leads to an increase of Smad1/5-regulated transcription. As a result, FGF-activated CaN signaling opposes BMP signaling during gastrulation, thereby promoting neural induction and the development of anterior structures.
Development of the nervous system begins with neural induction, which is controlled by complex signaling networks functioning in concert with one another. Fine-tuning of the bone morphogenetic protein (BMP) pathway is essential for neural induction in the developing embryo. However, the molecular mechanisms by which cells integrate the signaling pathways that contribute to neural induction have remained unclear. We find that neural induction is dependent on the Ca(2+)-activated phosphatase calcineurin (CaN). Fibroblast growth factor (FGF)-regulated Ca(2+) entry activates CaN, which directly and specifically dephosphorylates BMP-regulated Smad1/5 proteins. Genetic and biochemical analyses revealed that CaN adjusts the strength and transcriptional output of BMP signaling and that a reduction of CaN activity leads to an increase of Smad1/5-regulated transcription. As a result, FGF-activated CaN signaling opposes BMP signaling during gastrulation, thereby promoting neural induction and the development of anterior structures.
Zhang Y, Pak C, Han Y, Ahlenius H, Zhang Z, Chanda S, Marro S, Patzke C, Acuna C, Covy J, Xu W, Yang N, Danko T, Chen L, Wernig M, Südhof TC
Available methods for differentiating human embryonic stem cells (ESCs) and induced pluripotent cells (iPSCs) into neurons are often cumbersome, slow, and variable. Alternatively, human fibroblasts can be directly converted into induced neuronal (iN) cells. However, with present techniques conversion is inefficient, synapse formation is limited, and only small amounts of neurons can be generated. Here, we show that human ESCs and iPSCs can be converted into functional iN cells with nearly 100% yield and purity in less than 2 weeks by forced expression of a single transcription factor. The resulting ES-iN or iPS-iN cells exhibit quantitatively reproducible properties independent of the cell line of origin, form mature pre- and postsynaptic specializations, and integrate into existing synaptic networks when transplanted into mouse brain. As illustrated by selected examples, our approach enables large-scale studies of human neurons for questions such as analyses of human diseases, examination of human-specific genes, and drug screening.
Available methods for differentiating human embryonic stem cells (ESCs) and induced pluripotent cells (iPSCs) into neurons are often cumbersome, slow, and variable. Alternatively, human fibroblasts can be directly converted into induced neuronal (iN) cells. However, with present techniques conversion is inefficient, synapse formation is limited, and only small amounts of neurons can be generated. Here, we show that human ESCs and iPSCs can be converted into functional iN cells with nearly 100% yield and purity in less than 2 weeks by forced expression of a single transcription factor. The resulting ES-iN or iPS-iN cells exhibit quantitatively reproducible properties independent of the cell line of origin, form mature pre- and postsynaptic specializations, and integrate into existing synaptic networks when transplanted into mouse brain. As illustrated by selected examples, our approach enables large-scale studies of human neurons for questions such as analyses of human diseases, examination of human-specific genes, and drug screening.
Chanda S, Marro S, Wernig M, Südhof TC
Recent studies suggest that induced neuronal (iN) cells that are directly transdifferentiated from nonneuronal cells provide a powerful opportunity to examine neuropsychiatric diseases. However, the validity of using this approach to examine disease-specific changes has not been demonstrated. Here, we analyze the phenotypes of iN cells that were derived from murine embryonic fibroblasts cultured from littermate wild-type and mutant mice carrying the autism-associated R704C substitution in neuroligin-3. We show that neuroligin-3 R704C-mutant iN cells exhibit a large and selective decrease in AMPA-type glutamate receptor-mediated synaptic transmission without changes in NMDA-type glutamate receptor- or in GABAA receptor-mediated synaptic transmission. Thus, the synaptic phenotype observed in R704C-mutant iN cells replicates the previously observed phenotype of R704C-mutant neurons. Our data show that the effect of the R704C mutation is applicable even to neurons transdifferentiated from fibroblasts and constitute a proof-of-concept demonstration that iN cells can be used for cellular disease modeling.
Recent studies suggest that induced neuronal (iN) cells that are directly transdifferentiated from nonneuronal cells provide a powerful opportunity to examine neuropsychiatric diseases. However, the validity of using this approach to examine disease-specific changes has not been demonstrated. Here, we analyze the phenotypes of iN cells that were derived from murine embryonic fibroblasts cultured from littermate wild-type and mutant mice carrying the autism-associated R704C substitution in neuroligin-3. We show that neuroligin-3 R704C-mutant iN cells exhibit a large and selective decrease in AMPA-type glutamate receptor-mediated synaptic transmission without changes in NMDA-type glutamate receptor- or in GABAA receptor-mediated synaptic transmission. Thus, the synaptic phenotype observed in R704C-mutant iN cells replicates the previously observed phenotype of R704C-mutant neurons. Our data show that the effect of the R704C mutation is applicable even to neurons transdifferentiated from fibroblasts and constitute a proof-of-concept demonstration that iN cells can be used for cellular disease modeling.
Wapinski OL, Vierbuchen T, Qu K, Lee QY, Chanda S, Fuentes DR, Giresi PG, Ng YH, Marro S, Neff NF, Drechsel D, Martynoga B, Castro DS, Webb AE, Südhof TC, Brunet A, Guillemot F, Chang HY, Wernig M
Direct lineage reprogramming is a promising approach for human disease modeling and regenerative medicine, with poorly understood mechanisms. Here, we reveal a hierarchical mechanism in the direct conversion of fibroblasts into induced neuronal (iN) cells mediated by the transcription factors Ascl1, Brn2, and Myt1l. Ascl1 acts as an "on-target" pioneer factor by immediately occupying most cognate genomic sites in fibroblasts. In contrast, Brn2 and Myt1l do not access fibroblast chromatin productively on their own; instead, Ascl1 recruits Brn2 to Ascl1 sites genome wide. A unique trivalent chromatin signature in the host cells predicts the permissiveness for Ascl1 pioneering activity among different cell types. Finally, we identified Zfp238 as a key Ascl1 target gene that can partially substitute for Ascl1 during iN cell reprogramming. Thus, a precise match between pioneer factors and the chromatin context at key target genes is determinative for transdifferentiation to neurons and likely other cell types.
Direct lineage reprogramming is a promising approach for human disease modeling and regenerative medicine, with poorly understood mechanisms. Here, we reveal a hierarchical mechanism in the direct conversion of fibroblasts into induced neuronal (iN) cells mediated by the transcription factors Ascl1, Brn2, and Myt1l. Ascl1 acts as an "on-target" pioneer factor by immediately occupying most cognate genomic sites in fibroblasts. In contrast, Brn2 and Myt1l do not access fibroblast chromatin productively on their own; instead, Ascl1 recruits Brn2 to Ascl1 sites genome wide. A unique trivalent chromatin signature in the host cells predicts the permissiveness for Ascl1 pioneering activity among different cell types. Finally, we identified Zfp238 as a key Ascl1 target gene that can partially substitute for Ascl1 during iN cell reprogramming. Thus, a precise match between pioneer factors and the chromatin context at key target genes is determinative for transdifferentiation to neurons and likely other cell types.
Yang N, Zuchero JB, Ahlenius H, Marro S, Ng YH, Vierbuchen T, Hawkins JS, Geissler R, Barres BA, Wernig M
Transplantation of oligodendrocyte precursor cells (OPCs) is a promising potential therapeutic strategy for diseases affecting myelin. However, the derivation of engraftable OPCs from human pluripotent stem cells has proven difficult and primary OPCs are not readily available. Here we report the generation of induced OPCs (iOPCs) by direct lineage conversion. Forced expression of the three transcription factors Sox10, Olig2 and Zfp536 was sufficient to reprogram mouse and rat fibroblasts into iOPCs with morphologies and gene expression signatures resembling primary OPCs. More importantly, iOPCs gave rise to mature oligodendrocytes that could ensheath multiple host axons when co-cultured with primary dorsal root ganglion cells and formed myelin after transplantation into shiverer mice. We propose direct lineage reprogramming as a viable alternative approach for the generation of OPCs for use in disease modeling and regenerative medicine.
Transplantation of oligodendrocyte precursor cells (OPCs) is a promising potential therapeutic strategy for diseases affecting myelin. However, the derivation of engraftable OPCs from human pluripotent stem cells has proven difficult and primary OPCs are not readily available. Here we report the generation of induced OPCs (iOPCs) by direct lineage conversion. Forced expression of the three transcription factors Sox10, Olig2 and Zfp536 was sufficient to reprogram mouse and rat fibroblasts into iOPCs with morphologies and gene expression signatures resembling primary OPCs. More importantly, iOPCs gave rise to mature oligodendrocytes that could ensheath multiple host axons when co-cultured with primary dorsal root ganglion cells and formed myelin after transplantation into shiverer mice. We propose direct lineage reprogramming as a viable alternative approach for the generation of OPCs for use in disease modeling and regenerative medicine.
Webb AE, Pollina EA, Vierbuchen T, Urbán N, Ucar D, Leeman DS, Martynoga B, Sewak M, Rando TA, Guillemot F, Wernig M, Brunet A
FOXO transcription factors are central regulators of longevity from worms to humans. FOXO3, the FOXO isoform associated with exceptional human longevity, preserves adult neural stem cell pools. Here, we identify FOXO3 direct targets genome-wide in primary cultures of adult neural progenitor cells (NPCs). Interestingly, FOXO3-bound sites are enriched for motifs for bHLH transcription factors, and FOXO3 shares common targets with the proneuronal bHLH transcription factor ASCL1/MASH1 in NPCs. Analysis of the chromatin landscape reveals that FOXO3 and ASCL1 are particularly enriched at the enhancers of genes involved in neurogenic pathways. Intriguingly, FOXO3 inhibits ASCL1-dependent neurogenesis in NPCs and direct neuronal conversion in fibroblasts. FOXO3 also restrains neurogenesis in vivo. Our study identifies a genome-wide interaction between the prolongevity transcription factor FOXO3 and the cell-fate determinant ASCL1 and raises the possibility that FOXO3's ability to restrain ASCL1-dependent neurogenesis may help preserve the neural stem cell pool.
FOXO transcription factors are central regulators of longevity from worms to humans. FOXO3, the FOXO isoform associated with exceptional human longevity, preserves adult neural stem cell pools. Here, we identify FOXO3 direct targets genome-wide in primary cultures of adult neural progenitor cells (NPCs). Interestingly, FOXO3-bound sites are enriched for motifs for bHLH transcription factors, and FOXO3 shares common targets with the proneuronal bHLH transcription factor ASCL1/MASH1 in NPCs. Analysis of the chromatin landscape reveals that FOXO3 and ASCL1 are particularly enriched at the enhancers of genes involved in neurogenic pathways. Intriguingly, FOXO3 inhibits ASCL1-dependent neurogenesis in NPCs and direct neuronal conversion in fibroblasts. FOXO3 also restrains neurogenesis in vivo. Our study identifies a genome-wide interaction between the prolongevity transcription factor FOXO3 and the cell-fate determinant ASCL1 and raises the possibility that FOXO3's ability to restrain ASCL1-dependent neurogenesis may help preserve the neural stem cell pool.
Davila J, Chanda S, Ang CE, Südhof TC, Wernig M
We and others have reported the successful conversion of human fibroblasts into functional induced neuronal (iN) cells; however the reprogramming efficiencies were very low. Robust reprogramming methods must be developed before iN cells can be used for translational applications such as disease modeling or transplantation-based therapies. Here, we describe a novel approach in which we significantly enhance iN cell conversion efficiency of human fibroblast cells by reprogramming under hypoxic conditions (5% O₂). Fibroblasts were derived under high (21%) or low (5%) oxygen conditions and reprogrammed into iN cells using a combination of the four transcription factors BRN2, ASCL1, MYT1L and NEUROD1. An increase in Map2 immunostaining was only observed when fibroblasts experienced an acute drop in O₂ tension upon infection. Interestingly, cells derived and reprogrammed under hypoxic conditions did not produce more iN cells. Approximately 100% of patched cells fired action potentials in low O₂ conditions compared to 50% under high O₂ growth conditions, confirming the beneficial aspect of reprogramming under low O₂. Further characterization showed no significant difference in the intrinsic properties of iN cells reprogrammed in either condition. Surprisingly, the acute drop in oxygen tension did not affect cell proliferation or cell survival and was not synergistic with the blockade of GSK3β and Smad-mediated pathways. Our results showed that lowering the O₂ tension at the initiation of reprogramming is a simple and efficient strategy to enhance the production of iN cells which will facilitate their use for basic discovery and regenerative medicine.
We and others have reported the successful conversion of human fibroblasts into functional induced neuronal (iN) cells; however the reprogramming efficiencies were very low. Robust reprogramming methods must be developed before iN cells can be used for translational applications such as disease modeling or transplantation-based therapies. Here, we describe a novel approach in which we significantly enhance iN cell conversion efficiency of human fibroblast cells by reprogramming under hypoxic conditions (5% O₂). Fibroblasts were derived under high (21%) or low (5%) oxygen conditions and reprogrammed into iN cells using a combination of the four transcription factors BRN2, ASCL1, MYT1L and NEUROD1. An increase in Map2 immunostaining was only observed when fibroblasts experienced an acute drop in O₂ tension upon infection. Interestingly, cells derived and reprogrammed under hypoxic conditions did not produce more iN cells. Approximately 100% of patched cells fired action potentials in low O₂ conditions compared to 50% under high O₂ growth conditions, confirming the beneficial aspect of reprogramming under low O₂. Further characterization showed no significant difference in the intrinsic properties of iN cells reprogrammed in either condition. Surprisingly, the acute drop in oxygen tension did not affect cell proliferation or cell survival and was not synergistic with the blockade of GSK3β and Smad-mediated pathways. Our results showed that lowering the O₂ tension at the initiation of reprogramming is a simple and efficient strategy to enhance the production of iN cells which will facilitate their use for basic discovery and regenerative medicine.
Lujan E, Wernig M
The experimental induction of specific cell fates in related or unrelated lineages has fascinated developmental biologists for decades. The evaluation of altered cell fates in response to ectopic expression during embryonic development has been a standard assay for interrogating gene function. However, until recently examples of cell lineage conversions were limited to closely related and primitive cell types. The induction of pluripotency in fibroblasts prominently highlighted that combinations of transcription factors can be extremely powerful and are much more effective than single genes. On the basis of this conclusion we previously identified transcription factor combinations that directly induce functional neuronal cells from mesodermal and endodermal cells. This work has evoked numerous additional studies demonstrating direct lineage conversion into neural and other lineages. Here, we review the generation of neural progenitor cells from fibroblasts, which is the newest addition to the arena of induced cell types. Surprisingly, two fundamentally different approaches have been taken to induce this cell type, one direct approach and another that involves the intermediate generation of a partially reprogrammed pluripotent state.
The experimental induction of specific cell fates in related or unrelated lineages has fascinated developmental biologists for decades. The evaluation of altered cell fates in response to ectopic expression during embryonic development has been a standard assay for interrogating gene function. However, until recently examples of cell lineage conversions were limited to closely related and primitive cell types. The induction of pluripotency in fibroblasts prominently highlighted that combinations of transcription factors can be extremely powerful and are much more effective than single genes. On the basis of this conclusion we previously identified transcription factor combinations that directly induce functional neuronal cells from mesodermal and endodermal cells. This work has evoked numerous additional studies demonstrating direct lineage conversion into neural and other lineages. Here, we review the generation of neural progenitor cells from fibroblasts, which is the newest addition to the arena of induced cell types. Surprisingly, two fundamentally different approaches have been taken to induce this cell type, one direct approach and another that involves the intermediate generation of a partially reprogrammed pluripotent state.
Vierbuchen T, Wernig M
During development, diverse cellular identities are established and maintained in the embryo. Although remarkably robust in vivo, cellular identities can be manipulated using experimental techniques. Lineage reprogramming is an emerging field at the intersection of developmental and stem cell biology in which a somatic cell is stably reprogrammed into a distinct cell type by forced expression of lineage-determining factors. Lineage reprogramming enables the direct conversion of readily available cells from patients (such as skin fibroblasts) into disease-relevant cell types (such as neurons and cardiomyocytes) or into induced pluripotent stem cells. Although remarkable progress has been made in developing novel reprogramming methods, the efficiency and fidelity of reprogramming need to be improved in order increase the experimental and translational utility of reprogrammed cells. Studying the mechanisms that prevent successful reprogramming should allow for improvements in reprogramming methods, which could have significant implications for regenerative medicine and the study of human disease. Furthermore, lineage reprogramming has the potential to become a powerful system for dissecting the mechanisms that underlie cell fate establishment and terminal differentiation processes. In this review, we will discuss how transcription factors interface with the genome and induce changes in cellular identity in the context of development and reprogramming.
During development, diverse cellular identities are established and maintained in the embryo. Although remarkably robust in vivo, cellular identities can be manipulated using experimental techniques. Lineage reprogramming is an emerging field at the intersection of developmental and stem cell biology in which a somatic cell is stably reprogrammed into a distinct cell type by forced expression of lineage-determining factors. Lineage reprogramming enables the direct conversion of readily available cells from patients (such as skin fibroblasts) into disease-relevant cell types (such as neurons and cardiomyocytes) or into induced pluripotent stem cells. Although remarkable progress has been made in developing novel reprogramming methods, the efficiency and fidelity of reprogramming need to be improved in order increase the experimental and translational utility of reprogrammed cells. Studying the mechanisms that prevent successful reprogramming should allow for improvements in reprogramming methods, which could have significant implications for regenerative medicine and the study of human disease. Furthermore, lineage reprogramming has the potential to become a powerful system for dissecting the mechanisms that underlie cell fate establishment and terminal differentiation processes. In this review, we will discuss how transcription factors interface with the genome and induce changes in cellular identity in the context of development and reprogramming.
Lujan E, Chanda S, Ahlenius H, Südhof TC, Wernig M
We recently showed that defined sets of transcription factors are sufficient to convert mouse and human fibroblasts directly into cells resembling functional neurons, referred to as "induced neuronal" (iN) cells. For some applications however, it would be desirable to convert fibroblasts into proliferative neural precursor cells (NPCs) instead of neurons. We hypothesized that NPC-like cells may be induced using the same principal approach used for generating iN cells. Toward this goal, we infected mouse embryonic fibroblasts derived from Sox2-EGFP mice with a set of 11 transcription factors highly expressed in NPCs. Twenty-four days after transgene induction, Sox2-EGFP(+) colonies emerged that expressed NPC-specific genes and differentiated into neuronal and astrocytic cells. Using stepwise elimination, we found that Sox2 and FoxG1 are capable of generating clonal self-renewing, bipotent induced NPCs that gave rise to astrocytes and functional neurons. When we added the Pou and Homeobox domain-containing transcription factor Brn2 to Sox2 and FoxG1, we were able to induce tripotent NPCs that could be differentiated not only into neurons and astrocytes but also into oligodendrocytes. The transcription factors FoxG1 and Brn2 alone also were capable of inducing NPC-like cells; however, these cells generated less mature neurons, although they did produce astrocytes and even oligodendrocytes capable of integration into dysmyelinated Shiverer brain. Our data demonstrate that direct lineage reprogramming using target cell-type-specific transcription factors can be used to induce NPC-like cells that potentially could be used for autologous cell transplantation-based therapies in the brain or spinal cord.
We recently showed that defined sets of transcription factors are sufficient to convert mouse and human fibroblasts directly into cells resembling functional neurons, referred to as "induced neuronal" (iN) cells. For some applications however, it would be desirable to convert fibroblasts into proliferative neural precursor cells (NPCs) instead of neurons. We hypothesized that NPC-like cells may be induced using the same principal approach used for generating iN cells. Toward this goal, we infected mouse embryonic fibroblasts derived from Sox2-EGFP mice with a set of 11 transcription factors highly expressed in NPCs. Twenty-four days after transgene induction, Sox2-EGFP(+) colonies emerged that expressed NPC-specific genes and differentiated into neuronal and astrocytic cells. Using stepwise elimination, we found that Sox2 and FoxG1 are capable of generating clonal self-renewing, bipotent induced NPCs that gave rise to astrocytes and functional neurons. When we added the Pou and Homeobox domain-containing transcription factor Brn2 to Sox2 and FoxG1, we were able to induce tripotent NPCs that could be differentiated not only into neurons and astrocytes but also into oligodendrocytes. The transcription factors FoxG1 and Brn2 alone also were capable of inducing NPC-like cells; however, these cells generated less mature neurons, although they did produce astrocytes and even oligodendrocytes capable of integration into dysmyelinated Shiverer brain. Our data demonstrate that direct lineage reprogramming using target cell-type-specific transcription factors can be used to induce NPC-like cells that potentially could be used for autologous cell transplantation-based therapies in the brain or spinal cord.
Batista LF, Pech MF, Zhong FL, Nguyen HN, Xie KT, Zaug AJ, Crary SM, Choi J, Sebastiano V, Cherry A, Giri N, Wernig M, Alter BP, Cech TR, Savage SA, Reijo Pera RA, Artandi SE
The differentiation of patient-derived induced pluripotent stem cells (iPSCs) to committed fates such as neurons, muscle and liver is a powerful approach for understanding key parameters of human development and disease. Whether undifferentiated iPSCs themselves can be used to probe disease mechanisms is uncertain. Dyskeratosis congenita is characterized by defective maintenance of blood, pulmonary tissue and epidermal tissues and is caused by mutations in genes controlling telomere homeostasis. Short telomeres, a hallmark of dyskeratosis congenita, impair tissue stem cell function in mouse models, indicating that a tissue stem cell defect may underlie the pathophysiology of dyskeratosis congenita. Here we show that even in the undifferentiated state, iPSCs from dyskeratosis congenita patients harbour the precise biochemical defects characteristic of each form of the disease and that the magnitude of the telomere maintenance defect in iPSCs correlates with clinical severity. In iPSCs from patients with heterozygous mutations in TERT, the telomerase reverse transcriptase, a 50% reduction in telomerase levels blunts the natural telomere elongation that accompanies reprogramming. In contrast, mutation of dyskerin (DKC1) in X-linked dyskeratosis congenita severely impairs telomerase activity by blocking telomerase assembly and disrupts telomere elongation during reprogramming. In iPSCs from a form of dyskeratosis congenita caused by mutations in TCAB1 (also known as WRAP53), telomerase catalytic activity is unperturbed, yet the ability of telomerase to lengthen telomeres is abrogated, because telomerase mislocalizes from Cajal bodies to nucleoli within the iPSCs. Extended culture of DKC1-mutant iPSCs leads to progressive telomere shortening and eventual loss of self-renewal, indicating that a similar process occurs in tissue stem cells in dyskeratosis congenita patients. These findings in iPSCs from dyskeratosis congenita patients reveal that undifferentiated iPSCs accurately recapitulate features of a human stem cell disease and may serve as a cell-culture-based system for the development of targeted therapeutics.
The differentiation of patient-derived induced pluripotent stem cells (iPSCs) to committed fates such as neurons, muscle and liver is a powerful approach for understanding key parameters of human development and disease. Whether undifferentiated iPSCs themselves can be used to probe disease mechanisms is uncertain. Dyskeratosis congenita is characterized by defective maintenance of blood, pulmonary tissue and epidermal tissues and is caused by mutations in genes controlling telomere homeostasis. Short telomeres, a hallmark of dyskeratosis congenita, impair tissue stem cell function in mouse models, indicating that a tissue stem cell defect may underlie the pathophysiology of dyskeratosis congenita. Here we show that even in the undifferentiated state, iPSCs from dyskeratosis congenita patients harbour the precise biochemical defects characteristic of each form of the disease and that the magnitude of the telomere maintenance defect in iPSCs correlates with clinical severity. In iPSCs from patients with heterozygous mutations in TERT, the telomerase reverse transcriptase, a 50% reduction in telomerase levels blunts the natural telomere elongation that accompanies reprogramming. In contrast, mutation of dyskerin (DKC1) in X-linked dyskeratosis congenita severely impairs telomerase activity by blocking telomerase assembly and disrupts telomere elongation during reprogramming. In iPSCs from a form of dyskeratosis congenita caused by mutations in TCAB1 (also known as WRAP53), telomerase catalytic activity is unperturbed, yet the ability of telomerase to lengthen telomeres is abrogated, because telomerase mislocalizes from Cajal bodies to nucleoli within the iPSCs. Extended culture of DKC1-mutant iPSCs leads to progressive telomere shortening and eventual loss of self-renewal, indicating that a similar process occurs in tissue stem cells in dyskeratosis congenita patients. These findings in iPSCs from dyskeratosis congenita patients reveal that undifferentiated iPSCs accurately recapitulate features of a human stem cell disease and may serve as a cell-culture-based system for the development of targeted therapeutics.
Pang ZP, Yang N, Vierbuchen T, Ostermeier A, Fuentes DR, Yang TQ, Citri A, Sebastiano V, Marro S, Südhof TC, Wernig M
Somatic cell nuclear transfer, cell fusion, or expression of lineage-specific factors have been shown to induce cell-fate changes in diverse somatic cell types. We recently observed that forced expression of a combination of three transcription factors, Brn2 (also known as Pou3f2), Ascl1 and Myt1l, can efficiently convert mouse fibroblasts into functional induced neuronal (iN) cells. Here we show that the same three factors can generate functional neurons from human pluripotent stem cells as early as 6 days after transgene activation. When combined with the basic helix-loop-helix transcription factor NeuroD1, these factors could also convert fetal and postnatal human fibroblasts into iN cells showing typical neuronal morphologies and expressing multiple neuronal markers, even after downregulation of the exogenous transcription factors. Importantly, the vast majority of human iN cells were able to generate action potentials and many matured to receive synaptic contacts when co-cultured with primary mouse cortical neurons. Our data demonstrate that non-neural human somatic cells, as well as pluripotent stem cells, can be converted directly into neurons by lineage-determining transcription factors. These methods may facilitate robust generation of patient-specific human neurons for in vitro disease modelling or future applications in regenerative medicine.
Somatic cell nuclear transfer, cell fusion, or expression of lineage-specific factors have been shown to induce cell-fate changes in diverse somatic cell types. We recently observed that forced expression of a combination of three transcription factors, Brn2 (also known as Pou3f2), Ascl1 and Myt1l, can efficiently convert mouse fibroblasts into functional induced neuronal (iN) cells. Here we show that the same three factors can generate functional neurons from human pluripotent stem cells as early as 6 days after transgene activation. When combined with the basic helix-loop-helix transcription factor NeuroD1, these factors could also convert fetal and postnatal human fibroblasts into iN cells showing typical neuronal morphologies and expressing multiple neuronal markers, even after downregulation of the exogenous transcription factors. Importantly, the vast majority of human iN cells were able to generate action potentials and many matured to receive synaptic contacts when co-cultured with primary mouse cortical neurons. Our data demonstrate that non-neural human somatic cells, as well as pluripotent stem cells, can be converted directly into neurons by lineage-determining transcription factors. These methods may facilitate robust generation of patient-specific human neurons for in vitro disease modelling or future applications in regenerative medicine.
Yang N, Ng YH, Pang ZP, Südhof TC, Wernig M
Cellular plasticity is a major focus of investigation in developmental biology. The recent discovery that induced neuronal (iN) cells can be generated from mouse and human fibroblasts by expression of defined transcription factors suggested that cell fate plasticity is much wider than previously anticipated. In this review, we summarize the most recent developments in this nascent field and suggest criteria to help define and categorize iN cells that take into account the complexity of neuronal identity.
Cellular plasticity is a major focus of investigation in developmental biology. The recent discovery that induced neuronal (iN) cells can be generated from mouse and human fibroblasts by expression of defined transcription factors suggested that cell fate plasticity is much wider than previously anticipated. In this review, we summarize the most recent developments in this nascent field and suggest criteria to help define and categorize iN cells that take into account the complexity of neuronal identity.
Sebastiano V, Maeder ML, Angstman JF, Haddad B, Khayter C, Yeo DT, Goodwin MJ, Hawkins JS, Ramirez CL, Batista LF, Artandi SE, Wernig M, Joung JK
The combination of induced pluripotent stem cell (iPSC) technology and targeted gene modification by homologous recombination (HR) represents a promising new approach to generate genetically corrected, patient-derived cells that could be used for autologous transplantation therapies. This strategy has several potential advantages over conventional gene therapy including eliminating the need for immunosuppression, avoiding the risk of insertional mutagenesis by therapeutic vectors, and maintaining expression of the corrected gene by endogenous control elements rather than a constitutive promoter. However, gene targeting in human pluripotent cells has remained challenging and inefficient. Recently, engineered zinc finger nucleases (ZFNs) have been shown to substantially increase HR frequencies in human iPSCs, raising the prospect of using this technology to correct disease causing mutations. Here, we describe the generation of iPSC lines from sickle cell anemia patients and in situ correction of the disease causing mutation using three ZFN pairs made by the publicly available oligomerized pool engineering method (OPEN). Gene-corrected cells retained full pluripotency and a normal karyotype following removal of reprogramming factor and drug-resistance genes. By testing various conditions, we also demonstrated that HR events in human iPSCs can occur as far as 82 bps from a ZFN-induced break. Our approach delineates a roadmap for using ZFNs made by an open-source method to achieve efficient, transgene-free correction of monogenic disease mutations in patient-derived iPSCs. Our results provide an important proof of principle that ZFNs can be used to produce gene-corrected human iPSCs that could be used for therapeutic applications.
The combination of induced pluripotent stem cell (iPSC) technology and targeted gene modification by homologous recombination (HR) represents a promising new approach to generate genetically corrected, patient-derived cells that could be used for autologous transplantation therapies. This strategy has several potential advantages over conventional gene therapy including eliminating the need for immunosuppression, avoiding the risk of insertional mutagenesis by therapeutic vectors, and maintaining expression of the corrected gene by endogenous control elements rather than a constitutive promoter. However, gene targeting in human pluripotent cells has remained challenging and inefficient. Recently, engineered zinc finger nucleases (ZFNs) have been shown to substantially increase HR frequencies in human iPSCs, raising the prospect of using this technology to correct disease causing mutations. Here, we describe the generation of iPSC lines from sickle cell anemia patients and in situ correction of the disease causing mutation using three ZFN pairs made by the publicly available oligomerized pool engineering method (OPEN). Gene-corrected cells retained full pluripotency and a normal karyotype following removal of reprogramming factor and drug-resistance genes. By testing various conditions, we also demonstrated that HR events in human iPSCs can occur as far as 82 bps from a ZFN-induced break. Our approach delineates a roadmap for using ZFNs made by an open-source method to achieve efficient, transgene-free correction of monogenic disease mutations in patient-derived iPSCs. Our results provide an important proof of principle that ZFNs can be used to produce gene-corrected human iPSCs that could be used for therapeutic applications.
Marius Wernig
wernig@stanford.edu
Dr. Marius Wernig is a Professor of Pathology and a Co-Director of the Institute for Stem Cell Biology and Regenerative Medicine at Stanford University. He graduated with an M.D. Ph.D. from the Technical University of Munich where he trained in developmental genetics in the lab of Rudi Balling. After completing his residency in Neuropathology and General Pathology at the University of Bonn, he then became a postdoctoral fellow in the lab of Dr. Rudolf Jaenisch at the Whitehead Institute for Biomedical Research/ MIT in Cambridge, MA. In 2008, Dr. Wernig joined the faculty of the Institute for Stem Cell Biology and Regenerative Medicine at Stanford University where he has been ever since.
He received an NIH Pathway to Independence Award, the Cozzarelli Prize for Outstanding Scientific Excellence from the National Academy of Sciences U.S.A., the Outstanding Investigator Award from the International Society for Stem Cell Research, the New York Stem Cell Foundation Robertson Stem Cell Prize, the Ogawa-Yamanaka Stem Cell Prize delivered by the Gladstone Institute and more recently has been named a Faculty Scholar by the Howard Hughes Medical Institute.
Dr. Wernig’s lab is interested in pluripotent stem cell biology and the molecular determinants of neural cell fate decisions. His laboratory was the first to generate functional neuronal cells reprogrammed directly from skin fibroblasts, which he termed induced neuronal (iN) cells. The lab is now working on identifying the molecular mechanisms underlying induced lineage fate changes, the phenotypic consequences of disease-causing mutations in human neurons and other neural lineages as well as the development of novel therapeutic gene targeting and cell transplantation-based strategies for a variety of monogenetic diseases.
Academic appointments
Associate Professor Institute for Stem Cell Biology and Regenerative Medicine
Member:
Bio-X
Cardiovascular Institute
Child Health Research Institute
Institute for Stem Cell Biology and Regenerative Medicine
Stanford Cancer Institute
Stanford Neurosciences Institute
Administrative appointments
Faculty Senate, Department of Pathology (2017 - Present)
Assistant Professor, Institute for Stem Cell Biology and Regenerative Medicine (2008 - 2014)
Honors & Awards
HHMI Faculty Scholar Award, Howard Hughes Medical Institute (2016)
New York Stem Cell Foundation Robertson Stem Cell Prize, New York Stem Cell Foundation (2014)
The Outstanding Young Investigator Award, International Society for Stem Cell Research (2013)
Ascina Award, Republic of Austria (2010)
Cozzarelli Prize for Outstanding Scientific Excellence, National Academy of Sciences USA (2009)
New Scholar in Aging, Ellison Medical Foundation (2010)
Robertson Investigator Award, New York Stem Cell Foundation (2010)
Donald E. and Delia B. Baxter Faculty Scholarship, Stanford University (2009)
Margaret and Herman Sokol Award, Biomedical Research (2007)
Longterm fellowship Human Frontiers Science Program Organisation, HFSP (2004-2006)
Boards, Advisory Committees
Professional Organizations Member, Society for Neuroscience (2003 - Present)
Member, International Society for Stem Cell Research (2004 - Present)
Editorial Board Member, Cell Stem Cell (2012 - Present)
Editorial Board Member, Stem Cell Reports (2013 - Present)
Member, Program Committee, Society for Neuroscience (2016 - Present)
Chair, Program Committee, International Society for Stem Cell Research (2017 - Present)
Professional Education
M.D., Technical University of Munich, Medicine (2000)
Team

I joined the Wernig lab as an LSRP in the summer of 2022 after graduating from UCLA. I’m interested in the regenerative capabilities of the human body and the molecular mechanisms underlying these regenerative systems, as well as the ways in which we can utilize these mechanisms to treat disease via tools such as cell and gene therapy. My research interests further include immunological topics such as cancer biology and the coevolution of pathogenic species and the immune response. In the Wernig lab, I am investigating methods of replacing dysfunctional microglia in the CNS in the context of neurodegenerative diseases, as well as the translational potential of gene editing and the role that genetically manipulated cells can play in treating such diseases. Outside the lab, I enjoy spending time at the beach, watching sports, and going on road trips with my friends.
Danwei Wu is a Stanford neurology resident in the Neuroscience Scholar Track and aspiring neuroimmunologist. Prior to starting residency, she completed an HHMI Medical Research Fellowship with Dr. Vann Bennett at Duke University studying neuron-specific membrane domains and their interaction with cytoskeletal structures. Her current research interest includes cell-based therapies for multiple sclerosis, molecular pathways of neuro-repair, and pathogenesis of autoimmunity. She is interested in developing new therapies for neurologic diseases. Outside of the lab, she enjoys hiking and reading science fiction.
I am a PhD student in the Neurosciences Interdepartmental Program and joined the lab in summer 2022. I am interested in developing methods to reprogram stem cells directly into oligodendrocytes. I then hope to use these cells to study the role of oligodendrocytes in aging and neurodegeneration. In my free time I like to read, ski, and play tennis.
I am a passionate cellular and molecular biologistwith expertise in research related to cancer, genomic/chromosomal instability, DNA damage response, epigenetics, proteomics and cellular identity. The latter topic attracted me to the Wernig lab where I aim to decipher the mechanisms that allow us to specifically switch the identity of a cell, converting differentiated somatic cells into induced neurons. I also work on a project that aims to integrate CRISPR/CAS9-mediated gene correction with iPS cell generation in order to establish a therapy for the devastating skin disease epidermolysis bullosa. In my free time I play underwater rugby, surf, spearfish and ski!
I am an undergraduate studying neurobiology and computer science. In the Wernig Lab, I am studying how the interactions between reprogramming factors and chromatin modifiers allow for fully developed fibrojacklynl@stanford.edublasts to reprogram into induced neuronal cells (iN).
I studied small GTPase signaling and how its perturbation can contribute to cancer and neurological disorders during my PhD training at Cold Spring Harbor Laboratory. Since then, I have become increasingly fascinated by the epigenetic mechanisms of gene regulation, and how changes in epigenome influence cell function and fate decisions. In the Wernig lab, I am currently investigating the interactions between the reprogramming factors and chromatin modifiers. More specifically, I am interested in finding out how such interactions enable a terminally differentiated cell—for example, a fibroblast—to acquire new transcriptional program that allows its reprogramming into induced neuron (iN).
I am a Bachelor of Science candidate in Bioengineering at Stanford University. My research in the Wernig Lag involves investigating the relationship between microglia and the IKBKAP gene, specifically its role in mechanisms such as microglia regeneration and the transdifferentiation of hematopoietic stem and progenitor cells into microglia-like cells.
Kayla is a PhD student in the Neurosciences Interdepartmental Program and joined the lab in summer 2022. She is interested in leveraging the lab's cell reprogramming techniques to study the roles of individual cell types in proteinopathies. She aims to faithfully recapitulate disease states and uncover the underlying molecular mechanisms that lead to neurodegeneration. In her free time, Kayla can be found birding, hiking, or performing with the Stanford Symphony Orchestra.
I received my BA in molecular and cell biology (Neurobiology focus) and computer science from UC Berkeley. I went on to do my PhD at Duke University, where I worked on engineering electrical synapses for precise neuromodulation in Kafui Dzirasa’s lab. Now, as a postdoc in Marius Wernig lab, I am interested in delving deeper into cellular engineering to better understand neuronal induction, synapse formation, and deliver treatment across the blood brain barrier using engineered microglia. In my free time, I enjoy cooking and coding/electrical engineering projects.
As a neurosurgical resident, my research interests include translational topics such as cell therapy and neuroregenerative mechanisms. In the Wernig lab, I study the cellular reprogramming potential of microglia. Moreover, I’m interested in the functional integration of induced neurons into neural systems.
I am an undergraduate student studying Human Biology. At Wernig Lab, I am helping to research how microglial cells organize and communicate with each other. I am interested in understanding the relationship between changes in the organization of microglia and other neural cells and the progression of neuronal diseases.
I received my Ph.D. in Neuroscience and Brain Technologies in 2021 from the University of Genoa and the Italian Institute of Technology, Italy. I joined the Wernig lab in December 2022. Here, I am interested in understanding how the microglia-derived factors end up in the neuronal lysosomes. Furthermore, I am interested in understanding how the accumulation and breakdown of these factors lead to neurodegenerative disorders using human induced pluripotent stem cell culture. I read non-fiction novels, sketch, and go for hikes in my free time.
Rida Rehman received her doctorate in Natural Sciences (Dr. Rer. Nat.), specializing in brain trauma and rehabilitation at Universtät Ulm, Germany in 2023. She also holds an M.S. in Biomedical Sciences and a B.S. in Applied Biosciences from NUST, Pakistan. Rida’s research has primarily focused on understanding the critical role of microglia in neurotrauma, degeneration, reprogramming and repair. Rida joined Wernig lab in 2023 and is primarily working on microglia to neuron reprogramming.
I worked as a neurologist for 10 years in Japan. During clinical practice, I saw a lot of patients who could not be treated by the current medicine. As a result, I became strongly attracted to basic research of neuroscience. I received my PhD in Dr. Tatsushi Toda’s lab at Kobe University, where I studied disease-modifying drug for Parkinson’s disease. As a post doc fellow in the Wernig lab, I’m interested in investigating the ubiquitylation enzymes that are associated with the pathophysiology of Alzheimer’s disease in order to cure patients with neurodegenerative diseases in the future. In my free time, I play with my children, go cycling, and read comics.
I am a PhD student in the Department of Neurosciences and I joined the Wernig lab in the summer of 2020. I hope to study the fundamental cell biology of microglia giving rise to stable tiling in the brain. Furthermore, I am interested in how these mechanisms contribute to brain homeostasis and change with neuronal disease.
During my PhD in Yongjuan Zhao and Honcheung Lee’s lab, I focused on studying the enzymatic activity and function of ADP-ribosyl cyclase, particularly the activity of SARM1 and its role in axon degeneration. This work sparked my interest in neuroscience and neuropathology. Microglia, the resident immune cells in the brain, play critical roles in neuronal function and can be either beneficial or detrimental in the context of neurodegenerative diseases.In Wernig’s lab, I am interested in investigating the function of microglia in aging and Alzheimer’s disease. My current research focuses on understanding the effects of clonal hematopoietic mutations on microglia and their role in the development of Alzheimer’s disease.
I'm excited about diving into the mysteries of cell fate changes at both the cellular and molecular biology levels, using both experimental and computational tools. In the Wernig lab, I'm on a fascinating journey exploring methods to reprogramm cells into specific neuronal fates. Plus, I'm eager to unravel the basic transcriptional regulation codes in these processes.
Alumni





.gif)

Alan Napole was an LSRP working on elucidating the mechanisms of transgene silencing following iN differentiation from pluripotent stem cells. Concurrently, Alan is also working on understanding the role of microglia in multiple sclerosis via EAE mice models. Previously, he was a CIRM scholar working closely with microglia replacement strategies to develop a human mouse microglia chimera for investigation of neuroimmunological diseases. Alan is currently applying to medical school with interests in neuroscience and surgery. Outside of the lab, he enjoys attending music festivals, playing soccer, and cooking. He moved on to grad school in his home town LA. We wish him all the best for his future endeavors!
Angel earned his B.S. in Biology at California State Polytechnic University, Pomona. As an undergraduate he studied the effects of nicotine on diabetic individuals. As a CIRM fellow at Stanford University, he is interested in using microglia cell replacement and stem cell technology as a potential therapeutic for neurological disorders. Unfortunately, COVID19 cut some of the last of his time he was planning to stay with us. We are proud of Angel that he got into the PhD program at UC Irvine where he moved during these challenging times of natural disasters of the year 2020. Good luck Angel!
Bahareh's research interests were focused on epigenetic regulation of cognition at the level of synapses, using iN system. In my free time she likes to do sports and outdoor activities, cook healthy food, make mixed drinks, play music, and collect vinyl records. After graduating from our Stem Cell and Regenerative Medicine PhD program she moved on as postdoctoral fellow to Boston.
Cheen Euong Ang was a bioengineering PhD student in the Wernig lab. He spent his undergraduate career at McGill University, graduating with a B.Sc. in Chemistry with a focus in biological chemistry. Since coming to Stanford, Cheen Euong has been working on investigating the mechanisms of iN reprogramming and applying the iN platform in disease modeling and aging. Other than doing research in the lab, Cheen Euong has participated several scientific outreach programs such as the Stanford Biomedical Engineering Society undergraduate academic mentoring program and Stanford Institute of Medical Summer High School Student Research Program. Cheen moved on to do his postdoc in Xiaowei Zhuang at Harvard University in Cambridge, MA.
Christian's project focused on the function of synaptic proteins in the context of human neurons. Brain research has made tremendous advances in the last decades using animal models. However, most neuropathological conditions, such as autism or schizophrenia, are unique to human physiology. Thus, I study the effect of genetic alterations of synaptic proteins in induced human neurons using immunohistochemistry, analysis of gene and protein expression, as well as electrophysiology. Christian moved on to a senior lecturer position at the newly formed university of Linz, Austria. Good luck there!
Graduating from UC Berkeley majoring in Biology with an honors in Neuroscience, and having worked as an MD at the Royal Melbourne Hospital, I have developed a particular interest in translational neuroscience. In the lab, I am investigating the efficacy of cell therapies for neurological disorders. I am currently working on microglia based therapies for major neurological diseases, including multiple sclerosis.
I am a molecular biologist with a focus on neuro- and cancer biology. I am interested in using human iPSCs to develop new in vitro disease models, particularly for neuropsychiatric disorders and neurooncology. This led me to the Wernig lab where I started my postdoc to explore the advantages of direct neuronal reprogramming and the intricacies of step-wise differentiation of neural cell types. I am genetically engineering iPSC lines to generate complex genomic alterations and inducible mutations. Following differentiation into the corresponding disease-relevant tissue, I am puzzling together cellular phenotypes, transcriptional regulation, protein interactions, and epigenetic changes for a better understanding of the disease biology. In my spare time, I enjoy a new definition of chaos by my 2-year-old daughter.
Hiroko received Ph.D. from UCLA Neuroscience program in Jim Waschek's lab. She's currently a postdoc studying the disease mechanism of pediatric brain disorder Pelizaeus-Merzbacher Disorder, affecting oligodendrocyte development and myelination. She uses patient-derived iPS cells, gene targeting in iPS cells, and animal models.
I was an administrative associate for the Wernig Lab for a brief period in 2019. I am a native San Franciscan and I love the bay area. I received my undergrad from San Francisco State. I am fluent inSpanish so if you ever need a Spanish lesson come by desk. My husband and I have three beautiful childrenand during my time off I really enjoying spending time with my family.
I grew up in Long Island, NY. I attended college at Cornell University majoring in Biological Sciences with a concentration in Neurobiology and Behavior. After graduation in 2009, I moved to Nashville, TN to join the Medical Scientist Training Program at Vanderbilt University where I earned a combined MD/PhD. In 2016, I started my residency in Neurosurgery at Stanford. In the Wernig lab, I am interested in developing microglia-based regenerative therapies for neurodevelopmental and neurodegenerative disorders.
I received my PhD from National Institute of Biological Sciences (NIBS) at Beijing, China. My PhD mentor is Dr. Xiaodong Wang, who is a chinese-born American biochemist best known for his work with cytochrome C on apoptosis. He is a member of United States National Academy of Sciences and Howard Hughes Medical Institute.
During my PhD stage, my major work is to try and find function of a protein named RIP3 in necrosis (other program cell death) pathway. I use human fetal neural stem cells to study mutation caused epigenetic rigidity in reprogramming process. A lot of patient diseases are caused by gene mutation, but we do not know which kind of cells and which site of mutate will cause disease. We try to work on neural stem cells to induce some point mutation to imitate brain cancer disease from reported mutation site in human patient. Then we will know which mutation site is the key and try to fix it by the biology method.
The immense potential of genetic engineering technology as a tool to understand disease mechanisms and as a therapy motivates me to pursue research. I am studying CRISPR/Cas9 mediated gene correction of mutations in patients suffering from Epidermolysis bullosa. I am also studying iPSC-derived induced neurons to model neurological disorders.
Minjeong was the administrative associate for the Wernig Lab. We all loved her positive spirit that made our lab a happy place. We were fortunate to have her for a full year! During her time off, she loved hanging out with my family and trying out different recipes. She moved on to a full time position in business.
Dr. Moritz Mall is a postdoctoral fellow in the lab. After studying biochemistry and molecular biology at the LMU in Munich and the ETH in Zurich he received his Ph.D. from the EMBL in Heidelberg for his mechanistic studies on mitotic cell division. Besides surfing Moritz’s passion is to understand the molecular mechanisms of cell fate determination during reprogramming, development and disease.
A striking feature of the mammalian nervous system is its enormous cellular diversity. The biological question that drives my research in long-term is to understand how the nervous system develops and achieves its extraordinary complexity. I would also like to leverage my expertise in lineage reprogramming, stem cell biology, and neurobiology to develop reprogramming-based human cell culture models and study the fundamental processes underlying the development and function of human nervous system under normal and pathological conditions. Outside the lab, I like spending my time cycling, rock climbing and hiking.
I grew up in Australia where I completed an undergraduate degree in neuroscience and an MD at the University of Melbourne, Australia. I then completed my intern year at the Royal Melbourne Hospital. I have a keen interest in stem cell therapeutics and their potential application in treating debilitating neurological conditions. My current research focus is on novel microglia based therapies that can potentially ameliorate disease progression in multiple sclerosis.
With billions of neurons interconnected with many billions of synapses, our brain is the most complex object in the known universe. I like to think that I’m doing my part in understanding it. I use human neurons to study a synaptic protein called Neuroligin 4 that is not found in mice, therefore very complicated to study but at the same time extremely fascinating. When mutated Neuroligin 4 causes Autism and impairs the synaptic transmission of neurons. After leaving his prominent imprint on the lab he moved on to join the faculty at Mount Sanai School of Medicine in New York. Good luck Samuele, we will miss you!
My research interest is to investigate how genetic background contributes to the pathomechanism of complex mental disorders such autism or schizophrenia. In order to achieve my goals I am using various experimental techniques including rodent and human neuronal cell culture preparations, immunohistochemistry, gene and protein expression analysis and electrophysiology. As a scientist, my motto is: "We work in the dark to serve the light." Tamas moved back to his home country Hungary.
Virginia is a dentist that worked with underserved communities before taking time off to care for her family. She is happy to be the administrative associate for the Wernig lab. In her free time she enjoys reading, playing tennis, and spending time with her family. Unfortunately, due to COVID19 Virginia needed more time to care for her children and had to leave us. She was a delight to have around and we will dearly miss her!
Wendy received her Ph.D. from Columbia University, where she studied how cholesterol-rich membrane microdomains regulate a distinct function of a transmembrane protein, resulting in specific behavioral outcomes. In the Wernig lab, she aimed to understand the molecular mechanisms by which neuronal identity is established and how its disruption leads to neurological diseases focusing on the gene Myt1l.
Yingfei Liu is a Neurobiology PhD candidate from Xi`an Jiaotong University,China. She is studying here for two years as a Joint-Training Doctoral Program. Her previous research interest was the self-renewal and differentiation of the neural stem cells. Now She is studying the mechanisms of direct lineage reprogramming of fibroblasts into neurons. Yingfei was great to have in the lab. COVID19 also affected Yingfei's carreer. In the midst of great progress COVID19 precluded much of her continuing to work. She went back to China to arrange for her PhD defense and we are hoping to be able to continue to work with her in the future. Good luck Yingfei!
I completed my PhD in Dr. TY Chang’s lab at Dartmouth College, where I studied cholesterol metabolism in health and disease. In the Wernig lab, my research focus is on studying neurological diseases in human neurons and other neural lineages. I am also interested in developing novel therapeutic approaches for treating incurable neurological disorders using reprogramming technology.
Yongjin received his Ph.D. in functional genomics in 2018 from the Seoul National University, South Korea. For his Ph.D., he focused on genetic mutations of human neurological patients and its functional impacts. Yongjin joined the Wernig lab in October 2018. In the Wernig lab, he is interested in developing therapeutic methods for neurological patients and understanding disease mechanisms using human stem cells and neurons. We are proud that Yongjin accepted an offer to join the faculty at the prestigious Seoul University.
Press
Wernig Lab Protocols & Recipes
Contact
We are always interested to hear from ambitious scientists and potential collaborators.
Marius Wernig
wernig@stanford.edu
Amy Lang, Lab Admin
alang2@stanford.edu
Institute for Stem Cell Biology and Regenerative Medicine
Wernig Laboratory
265 Campus Drive G3141
Stanford, CA 94305
U.S.A.

